Side Effects After Switching to Generics: When to Worry and What to Do
 May, 4 2026
May, 4 2026
You pick up your prescription, expecting the same pill you’ve taken for months. Instead, it’s a different color or shape. The pharmacist says it’s just a generic drug with the same active ingredient as the brand-name version but potentially different inactive ingredients and manufacturing processes. You take it, and suddenly, you feel off. Your anxiety spikes, your thyroid levels fluctuate, or your seizures return. Is this in your head? Or is there something real happening?
The short answer is yes, it can be real. While regulatory agencies insist that generics are equivalent to brand-name drugs, clinical reality tells a more complex story. For most people, switching causes no issues. But for a significant minority-especially those on sensitive medications-the switch can trigger adverse effects or reduced efficacy. Understanding when to worry and how to manage these changes is crucial for your health.
Why Generics Can Feel Different
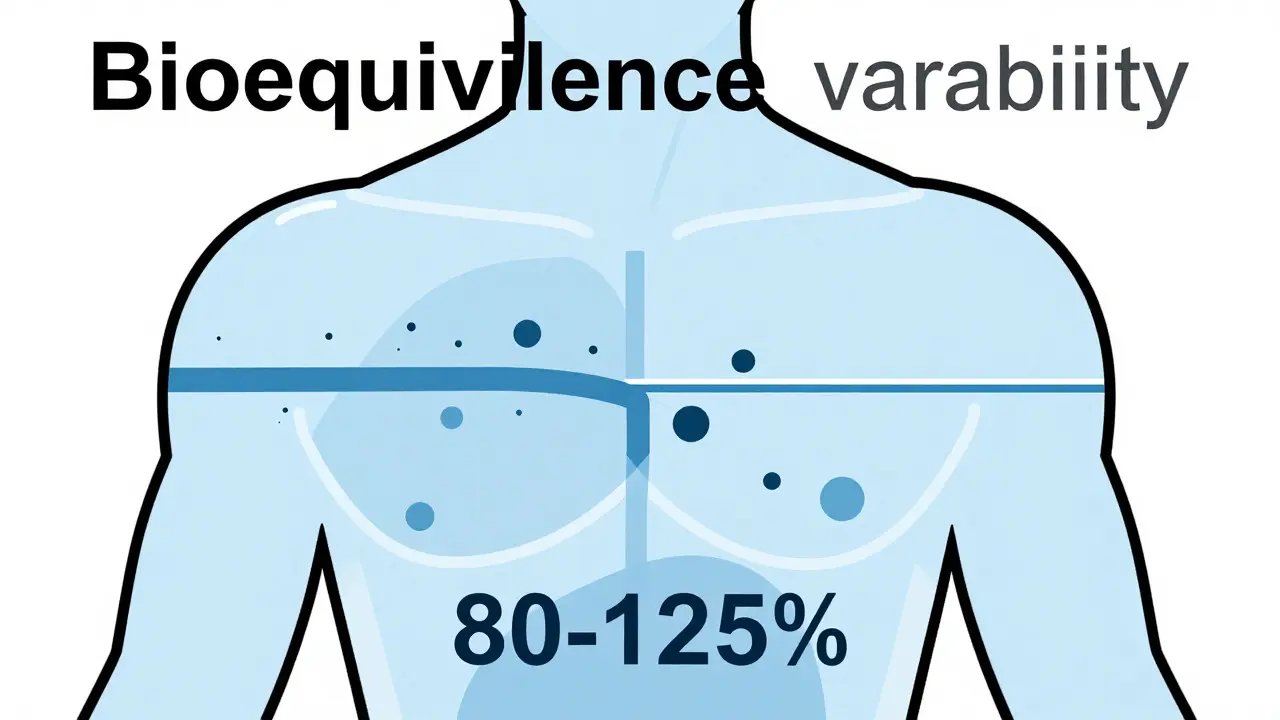
To understand why switching might bother you, we need to look at how generics are approved. In the United States, the FDA requires generics to demonstrate bioequivalence, meaning they must achieve 80-125% of the brand-name drug's pharmacokinetic profile in terms of absorption rate and extent. This range allows for up to a 20% variation in how fast and how much of the drug enters your bloodstream compared to the brand.
For most medications, this variance is clinically insignificant. Your body handles the slight differences without issue. However, this standard applies to average population responses. It does not account for individual variations. Some people have unique metabolic profiles or sensitivities that make them react strongly to even small changes in drug delivery.
Furthermore, while the active ingredient must be identical, the inactive ingredients-fillers, binders, dyes, and coatings-can differ significantly between manufacturers. These excipients don’t treat your condition, but they affect how the drug dissolves and releases in your gut. A change in these components can alter the drug’s performance in ways that aren’t captured by standard bioequivalence testing.
High-Risk Medications: When Switching Matters Most
Not all drugs carry the same risk when switched. The danger lies primarily with medications that have a Narrow Therapeutic Index (NTI) has a very small difference between the dose that helps and the dose that harms, requiring precise blood concentration levels for safe and effective treatment. For NTI drugs, the FDA tightens the allowable difference to 10-15%, recognizing their heightened sensitivity. Even so, switching between different generic manufacturers of the same NTI drug can still cause problems because each manufacturer may use different release mechanisms.
Research identifies five key categories where switching concerns are most prevalent:
- Antiepileptics: Drugs like divalproex sodium and phenytoin require stable blood levels to prevent seizures. A 2021 survey found that nearly 70% of neurologists believe generic switches increase breakthrough seizure risk.
- Thyroid Medications: Levothyroxine is highly sensitive to formulation changes. Fluctuations can lead to symptoms of hypo- or hyperthyroidism, affecting energy, weight, and heart rate.
- Anticoagulants: Warfarin users need consistent dosing to avoid bleeding risks or clot formation. Variability in absorption can disrupt INR levels.
- Immunosuppressants: Tacrolimus and cyclosporine, used in transplant patients, have narrow margins for error. Rejection episodes can occur if drug levels drop unexpectedly.
- Psychiatric Medications: Extended-release formulations for ADHD (like Adderall XR) and certain antidepressants often show noticeable efficacy drops or new side effects after switching.
If you are taking any of these medications, you should pay close attention to who manufactures your pills. Consistency matters more here than cost savings.
Real-World Evidence: What Studies Show
It’s easy to dismiss anecdotal reports as placebo effects, but robust data supports patient experiences. A 2019 retrospective cohort study published in BMJ Open analyzed over 2,800 patients with cardiovascular disease. They found a 12.3% increased risk of hospitalization within 30 days of switching between generic products of the same active substance compared to non-switchers.
Another study focusing on ADHD medications revealed even starker contrasts. Dr. Lenard A. Guttman from the University of Southern California reported that 30-40% of his ADHD patients experienced clinically meaningful differences when switched between generic manufacturers. Symptoms returned or new side effects emerged within 24-72 hours. Similarly, a MedShadow survey found that 63.2% of patients noticed decreased efficacy after a manufacturer switch, with headaches, anxiety, and gastrointestinal issues being common complaints.
These findings contradict the official stance that generics are therapeutically equivalent for everyone. The tension exists because regulatory standards focus on statistical averages, while patients experience individual biological realities. If you fall outside the average, the "equivalent" label might not protect you.
Why Pharmacies Keep Changing Your Pills
You might wonder why your pharmacy keeps giving you different-looking pills every month. The answer isn’t medical; it’s economic. Pharmacy Benefit Managers (PBMs) operate systems that incentivize pharmacies to switch patients between generic manufacturers monthly based on which supplier offers the highest rebates. Companies like CVS Caremark and Express Scripts change preferred manufacturers an average of 4.7 times annually per drug.
This practice creates instability in medication supply. You might get Manufacturer A one month, Manufacturer B the next, and Manufacturer C the month after. Each switch carries a potential risk of adverse effects. For the 187 million Americans with prescription drug coverage, this constant churn affects millions of lives. While generics save billions in healthcare costs, the hidden costs of problematic switching-including estimated $2.1 billion annually in preventable hospitalizations-are increasingly recognized.
When to Worry: Signs That Something Is Wrong
How do you know if a side effect is due to the switch or just coincidence? Look for timing and pattern. Adverse effects from switching typically emerge within 24-72 hours of starting the new formulation. Common signs include:
- Return of original symptoms: If your depression lifts, then dips again after a refill, the new generic might be less effective.
- New physical side effects: Headaches, nausea, jitteriness, or insomnia that weren’t present before.
- Lab value fluctuations: Unexplained changes in TSH (thyroid), INR (blood thinners), or drug level monitoring tests.
- Behavioral changes: Increased anxiety, mood swings, or irritability.
If you notice these changes shortly after picking up a differently colored or shaped pill, document it immediately. Don’t assume it’s "just in your head." Bring this evidence to your doctor or pharmacist.
Practical Steps to Manage Generic Switches
You have power in this process. Here’s how to protect yourself:
- Check the NDC Code: Every prescription box has a National Drug Code (NDC). The first segment identifies the manufacturer. Write down the code from your current working pill. Compare it to new refills.
- Ask for Specific Manufacturers: Pharmacists can often accommodate requests for specific generic manufacturers if stock is available. Tell them which manufacturer works best for you.
- Use "Dispense as Written" Codes: For NTI drugs, ask your prescriber to mark the prescription as "DAW-1" (substitution not permitted). This forces the pharmacy to dispense the exact product specified, though it may increase your copay.
- Keep a Medication Log: Track manufacturer changes alongside any symptoms. This log provides concrete data for your doctor to review.
- Talk to Your Pharmacist: A 2021 study showed that patients receiving pharmacist-led counseling about switching were 37% less likely to experience adverse outcomes. Build a relationship with your local pharmacist.
Some health systems, like the University of Michigan Health System, have implemented "medication fingerprinting" programs that reduce switching-related adverse events by over 50%. You can advocate for similar consistency in your care.
Regulatory Changes and Future Outlook
Awareness of these issues is growing. The FDA launched its Complex Generic Products Initiative in 2020 to develop better testing methods for drugs where standard bioequivalence measures fail. In 2023, CMS issued guidance limiting generic manufacturer changes to twice annually for Medicare Part D plans, aiming to reduce adverse events by 31.4%.
However, implementation remains limited. Industry resistance persists due to the massive savings generics provide. As a patient, you must remain vigilant. Regulatory improvements will help, but personal advocacy remains essential right now.
| Feature | Brand-Name Drug | Generic Drug |
|---|---|---|
| Active Ingredient | Identical | Identical |
| Inactive Ingredients | Consistent across batches | May vary by manufacturer |
| Cost | Higher | Lower (often 80% less) |
| Bioequivalence Range | N/A (Reference) | 80-125% of brand reference |
| Switching Risk | Low | Variable (High for NTI drugs) |
Are generic drugs really the same as brand-name drugs?
They contain the same active ingredient and must meet bioequivalence standards (80-125% absorption range). However, inactive ingredients and manufacturing processes differ, which can affect how some individuals respond, especially with narrow therapeutic index drugs.
Why does my pill look different every month?
Pharmacies often switch between generic manufacturers based on rebate incentives from Pharmacy Benefit Managers (PBMs). This frequent switching can lead to variability in drug performance for sensitive patients.
What are Narrow Therapeutic Index (NTI) drugs?
NTI drugs have a very small margin between effective and toxic doses. Examples include levothyroxine, warfarin, and certain antiepileptics. Small changes in absorption can lead to serious health consequences, making manufacturer consistency critical.
Can I request a specific generic manufacturer?
Yes. You can ask your pharmacist for a specific manufacturer if available. Additionally, your doctor can write "Dispense as Written" (DAW-1) on the prescription to prevent substitution, though this may increase your out-of-pocket cost.
When should I contact my doctor about a generic switch?
Contact your doctor if you experience a return of original symptoms, new side effects like headaches or anxiety, or unexpected lab result fluctuations within 24-72 hours of starting a new generic formulation.
Do all generic switches cause problems?
No. For most patients and most drugs, generics work perfectly fine. Problems are more common with extended-release formulations, psychiatric meds, and narrow therapeutic index drugs. Individual sensitivity varies.
What is bioequivalence?
Bioequivalence means the generic drug reaches the bloodstream at a similar rate and extent as the brand-name drug. The FDA allows an 80-125% range, which is statistically equivalent for populations but may impact individual patients.
How can I track which manufacturer makes my pills?
Look at the National Drug Code (NDC) on your prescription bottle. The first segment identifies the manufacturer. Keep a log of these codes alongside any symptoms to identify patterns.
Srinivas Komakula
May 5, 2026 AT 05:32The bioequivalence range of 80-125% is not a statistical oversight; it is a deliberate mechanism of control. The regulatory bodies, in collusion with the PBM industrial complex, have engineered a system where 'therapeutic equivalence' is a legal fiction designed to maximize profit margins at the expense of physiological integrity. The excipients are not merely fillers; they are vectors for subtle neurotoxicity and endocrine disruption that standard pharmacokinetic models fail to capture because they are calibrated for the median population, not the individual outlier. We must recognize that the variance allowed is essentially a license for malpractice by design.
Preety Singh
May 5, 2026 AT 07:50One cannot simply dismiss the clinical reality of these fluctuations as anecdotal noise. The data presented regarding NTI drugs is irrefutable evidence of systemic negligence. It is rather pathetic that patients are expected to absorb the risks of corporate cost-cutting measures without recourse. The distinction between brand and generic is not merely semantic but biological. Ignoring this nuance is an act of intellectual laziness.
Seema Karanje
May 6, 2026 AT 03:12WAKE UP PEOPLE! You are being played like a fiddle while your health deteriorates! Stop accepting whatever garbage the pharmacy hands you! Demand better! Fight back! If you feel off, IT IS REAL! Do not let them gaslight you into thinking it's just in your head! Take charge of your body NOW!
J. Walter Jenkem
May 7, 2026 AT 07:17This is a very important discussion. I appreciate the clarity on the NDC codes. It seems many of us are unaware that we can actually track these changes. Collaboration between patients and pharmacists is key here. We need to support each other in navigating this confusing landscape.
Mark Koepsell
May 8, 2026 AT 06:01As a pharmacist, I can confirm that the rebate-driven switching is the primary driver here. We often receive shipments from different manufacturers based on the lowest bid, regardless of patient history. The DAW-1 code is underutilized. Patients should request it for NTI medications specifically. The clinical difference in levothyroxine absorption between manufacturers is measurable and significant for sensitive individuals.
Elizabeth Holden
May 9, 2026 AT 23:08u/8619 is right but most doctors dont even know this stuff. its crazy how much money pbms make off our suffering. i switched my adhd meds and felt like crap within days. everyone needs to check their ndc codes. its not hard. stop being lazy about your own health.
Jenny X
May 10, 2026 AT 18:05The narrative of 'bioequivalence' is a construct maintained by regulatory capture. The 80-125% window is a Trojan horse for pharmaceutical consolidation. When you consider the cumulative effect of excipient variation over decades, the long-term health implications are staggering. This is not a bug; it is a feature of the current healthcare architecture designed to shift liability onto the consumer.
bharat films
May 10, 2026 AT 21:14😡 This is so true! I had seizures return after a switch! 😱 The FDA is asleep at the wheel! 🛌💤 Wake up! ⚠️ Check your pills! 💊 Don't trust the big pharma machine! 🚫🏭 #GenericDrugs #HealthFreedom
Mikaela -anonymous 😏
May 12, 2026 AT 17:25Oh, how delightful. Another article telling us that our healthcare system is fundamentally broken. How original. One might think that after all these years, someone would have noticed that 'cost savings' usually means 'patient suffering.' But no, we must continue to applaud the efficiency of the machine while it grinds our health into dust. Bravo.
kelvin villa saab
May 12, 2026 AT 19:11look i am not a doctor but this makes sense. why would they change the pill if it works? greed. pure greed. i hate when they give me the blue one instead of the white one. it feels wrong. maybe we should just go back to home remedies or something. this whole system is messed up.
Spencer Farrell
May 14, 2026 AT 09:03The philosophical implication here is profound: the reductionist view of medicine fails to account for the holistic nature of human biology. To treat the body as a machine that accepts interchangeable parts is a category error. The variability in response highlights the uniqueness of individual existence against the homogenizing force of mass production. We must reject the false equivalence imposed by regulatory standards.
Kartik Agarwal
May 14, 2026 AT 13:26We must foster a community of informed advocacy. By leveraging the NDC tracking systems and demanding transparency from PBMs, we can create a feedback loop that forces accountability. The jargon surrounding bioequivalence is often used to obscure the clinical reality of therapeutic failure. Let us demystify these processes and empower ourselves through knowledge.
Kelly Feehely
May 15, 2026 AT 09:50Stop letting them push you around! These companies are thieves! They steal your health for pennies! I am sick of the excuses! If you are on warfarin or thyroid meds, YOU ARE A TARGET! Demand the brand name! Pay the extra money! Your life is worth more than their rebate! Fight now!